The Coronavirus lockdowns this spring disrupted many projects and students. But when life gives you lemons, make lemonade. In our lab, almost everyone took...
READ MORE
The Coronavirus lockdowns this spring disrupted many projects and students. But when life gives you lemons, make lemonade. In our lab, almost everyone took the stay-at-home orders as motivation to learn some coding. Sebastian Siegner, a Masters’ student at the time and now recently joined for a full Ph.D., had been working on proof-of-concept experiments for therapeutic base editing and prime editing. But he was frustrated with designing base editor gRNAs and newly-described prime editor pegRNAs by hand. So he used the lockdown to write PnB Designer, which is a fast and scalable helper to design your prime and base editing experiments. Check out the paper in BMC Bioinformatics.
PnB designer can be used to design base editing gRNAs and prime editing pegRNAs in both single-edit and batch mode. You can design against arbitrary DNA sequences (copy/paste your sequence as input), or you can enter genomic coordinates of your favorite gene in your favorite genome. Several species are currently supported, from human to plant. For base editors, just choose which nucleotide change you want to make and the software will take into account both the mutation and editing window to figure out the best editor to use. For prime editing, you can test all kinds of reverse transcriptase template (RTT) and primer binding site (PBS) lengths with a click of a button. The output is a table of possible gRNAs/pegRNAs, ordered by a heuristic score.
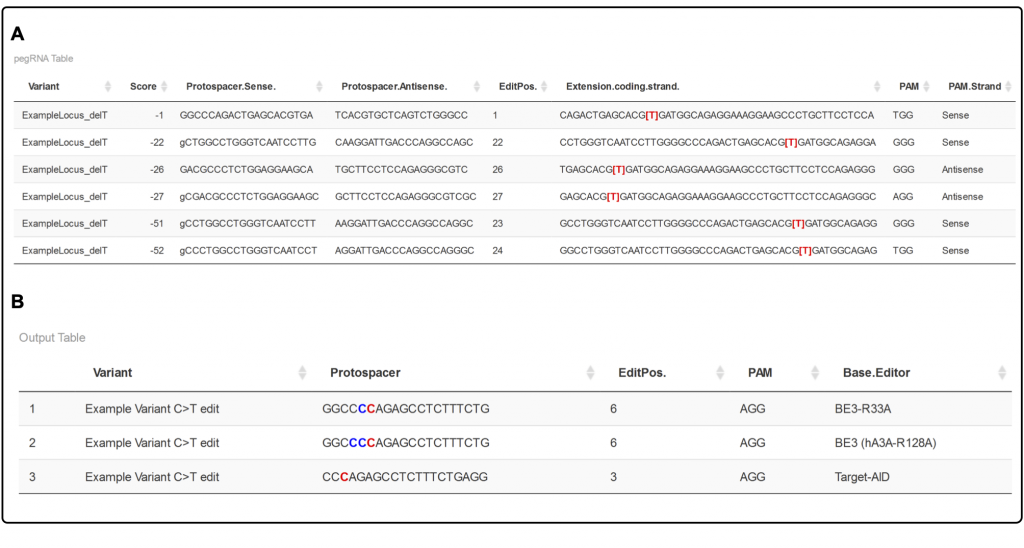
Sebastian tested PnB Designer by designing pegRNAs to model most of the human disease-associated mutations in ClinVar using prime editing. He even varied RTT and PBS length for each of these 96,000+ mutations, figuring out good parameters to keep pegRNAs at a reasonable length while still modelling ~80% of all variants.
This was a challenging but exciting side-project for Sebastian’s Masters’ degree. He wrote PnB Designer independently during the lockdown, and the rest of the lab acted as beta testers to provide suggestions. Congrats to Sebastian on your first paper, which is already being used by several people in the Zurich area. PnB Designer is completely free to use and is hosted by the Functional Genomics Center Zurich.
X Close


